Olympus Reduces Risk of Cross-Contamination and Costly Repairs with ALT-Pro Automatic Leak Tester for Endoscope Reprocessing
Fastest, Most Compact System Frees Up Staff Time and Counter Space,
and Can Prevent Human Error
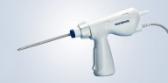
CENTER VALLEY, Pa., April 24, 2014 – Olympus, a global technology leader in delivering innovative diagnostic and therapeutic solutions for Medical and Surgical procedures, among other core businesses, announced today the launch of the fastest, most compact Automated Endoscope Leak Tester on the market, ALT-Pro – a system that prevents human error while enhancing efficiency in endoscope reprocessing.
“We’re pleased to introduce what may well be the gold standard in endoscope leak testing,” said Luke Calcraft, President of the Medical Systems Group at Olympus Corporation of the Americas. “The ALT-Pro’s innovative dry leak testing technology takes the guesswork out of endoscope leak testing by removing the human elements that lead to human error. With endoscopes serving as the channel for an expanding range of high-tech surgical and diagnostic devices, the technology used to protect them should also continue to advance. The ALT-Pro will be a welcome addition to reprocessing departments.”
Conventional endoscope leak testing includes pressurizing an endoscope with an air pumping device while submerging the endoscope in water, angulating the scope’s distal tip and manually flexing and manipulating the entire exterior area of the endoscope while observing the water for air bubbles. Submersion leak-testing methods rely heavily upon acute observation and proper technician training to accurately recognize early-stage endoscope leaks. By finding endoscope leaks sooner, the potential for endoscope cross-contamination and costly endoscope repairs can be reduced.
The ALT-Pro’s dry leak testing technology is fully computerized, creating a highly repeatable endoscope leak testing process for all skill levels. Simply attach up to two endoscopes, start the leak test process and walk away until the leak test is finished. A built-in Radio Frequency Identification (RFID) reader promotes accurate record keeping and enables precise early-stage leak detection by inflating the endoscope based on its specific model. Additionally, the ALT-Pro’s small footprint and optional wall mount reduce countertop clutter.
According to the American Society for Gastrointestinal Endoscopy, the risk of infection from scope contamination is one in 10 million procedures; however, the damage to one person’s health should not be underestimated, nor should the detriment a preventable infection can cause to one health facility’s brand.
ALT-Pro Benefits
The ALT-Pro dry endoscope leak test eliminates the need for endoscope submersion, air bubble observation and distal tip angulation. Other features include:
- Simple 3-step process: Scan (via RFID), Connect and Start
- Easy-to-read LCD display includes work flow instructions, test results and settings
- Choice between automated (dry) and conventional submersion leak testing modes to help meet your needs
- Self check on startup that ensures the unit is operating properly
- Electronic export feature that allows for records export to a PC for backup or analysis
- 12-month warranty that protects investment
The ALT-Pro helps address key requirements of Healthcare Reform and the Affordable Care Act, including:
- Increased Quality of Care – The ALT-Pro's fully automated dry leak
test process reduces human error by removing the human elements of manual endoscope leak testing methods. - Decreased Costs – The early detection of endoscope leaks may reduce
the severity of damage to the endoscopes’ internal components, reducing
expensive endoscope repair costs. - Enhanced Patient Satisfaction – The accurate detection of endoscope
leaks reduces the potential for patient infection through endoscope cross-
contamination.
The ALT-Pro will be showcased at the 2014 SGNA Annual Course to be held on May 2-6 in Nashville, Tenn. Healthcare providers are invited to visit Booth #400 to further explore the benefits of this important new leak testing technology. The ALT-Pro will also be showcased at APIC 2014 in Anaheim, Calif., June 7-9, 2014.
For more information or to evaluate the ALT-Pro, please contact your Olympus representative at 800-848-9024 or visit http://medical.olympusamerica.com/customer-resources/cleaning-disinfection-sterilization/reprocessing-products/alt-pro.
About Olympus Medical Systems Group
Olympus Medical Systems Group, a division of global technology leader Olympus, develops solutions for healthcare professionals that help improve clinical outcomes, reduce overall costs and enhance quality of life for their patients. By enabling less invasive procedures, innovative diagnostic and therapeutic endoscopy, and early stage lung cancer evaluation and treatments, Olympus is transforming the future of healthcare.
For more information visit Olympus at www.medical.olympusamerica.com.
Media Contact
Michael Levey
Olympus Corporation of the Americas
484-896-5120
michael.levey@olympus.com