
Ease the Squeeze with our Advanced Bipolar Vessel Sealing Portfolio
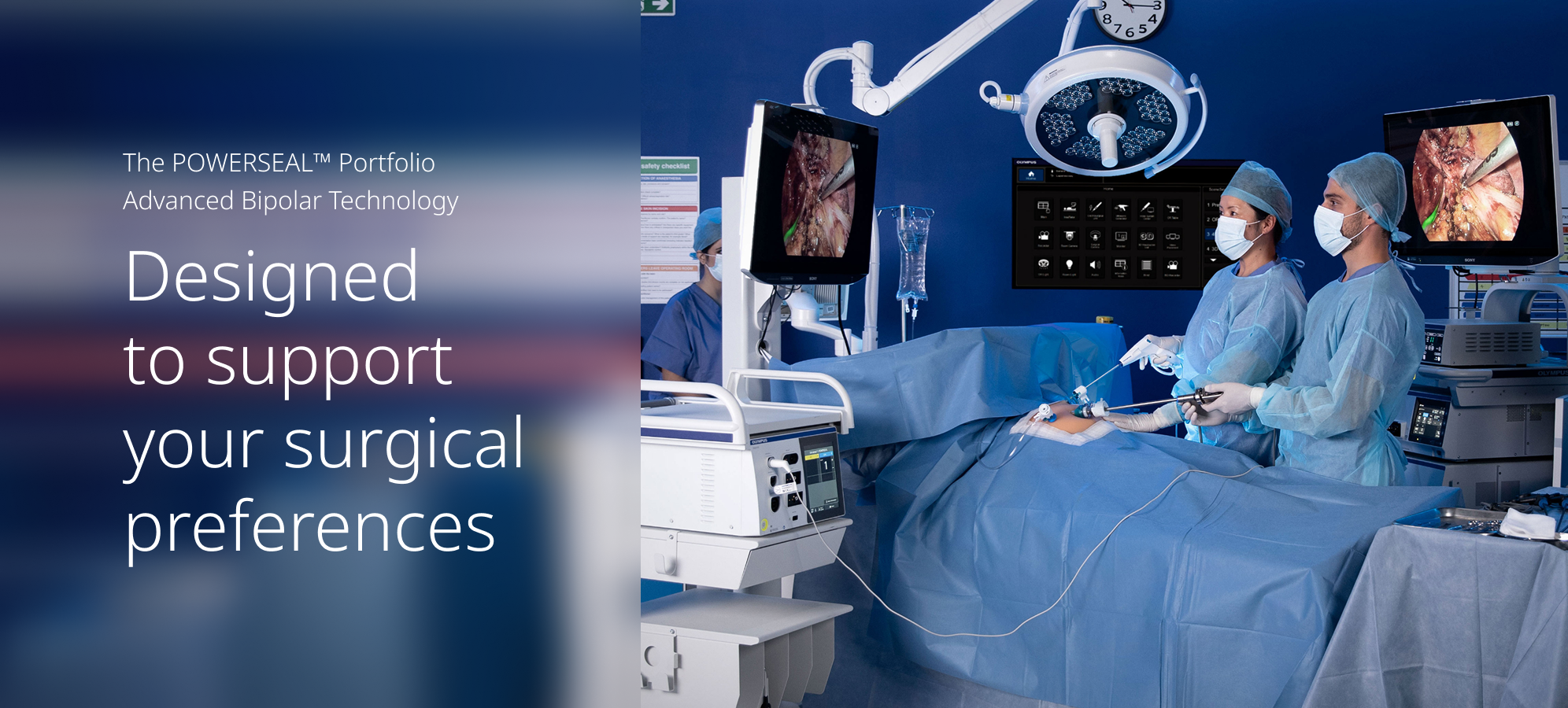
When the pressure is on, you need a surgical energy device that helps you stay in the flow and in control. The POWERSEAL™ device consistently delivers strong vessel sealing throughout the procedure1 and features an ergonomic handle designed to feel comfortable in your hand.2

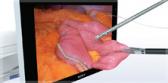
Confident Vessel Sealing
Efficient and reliable 7 mm vessel sealing with advanced bipolar technology.3
- Seals vessels up to and including 7 mm, including pulmonary vessels, tissue bundles, and lymphatics.3
- Secure sealing with a greater than 99% probability of vessel burst pressure greater than 360 mmHg.4
- Average seal times between 2.8 and 3.3 seconds.3
- Equivalent thermal spread when compared to other advanced bipolar vessel sealing devices.5
- Minimal tissue sticking due to unique waveform and jaw construction.6
Designed to fit naturally in your hand and deliver an enhanced ergonomic experience.
- Up to 66% less squeeze force required to close the jaws.2
- Reduced reach distances to device landmarks.2
- Latch-on option maintains secure jaw position while grasping and retracting or sealing vessels.


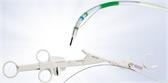
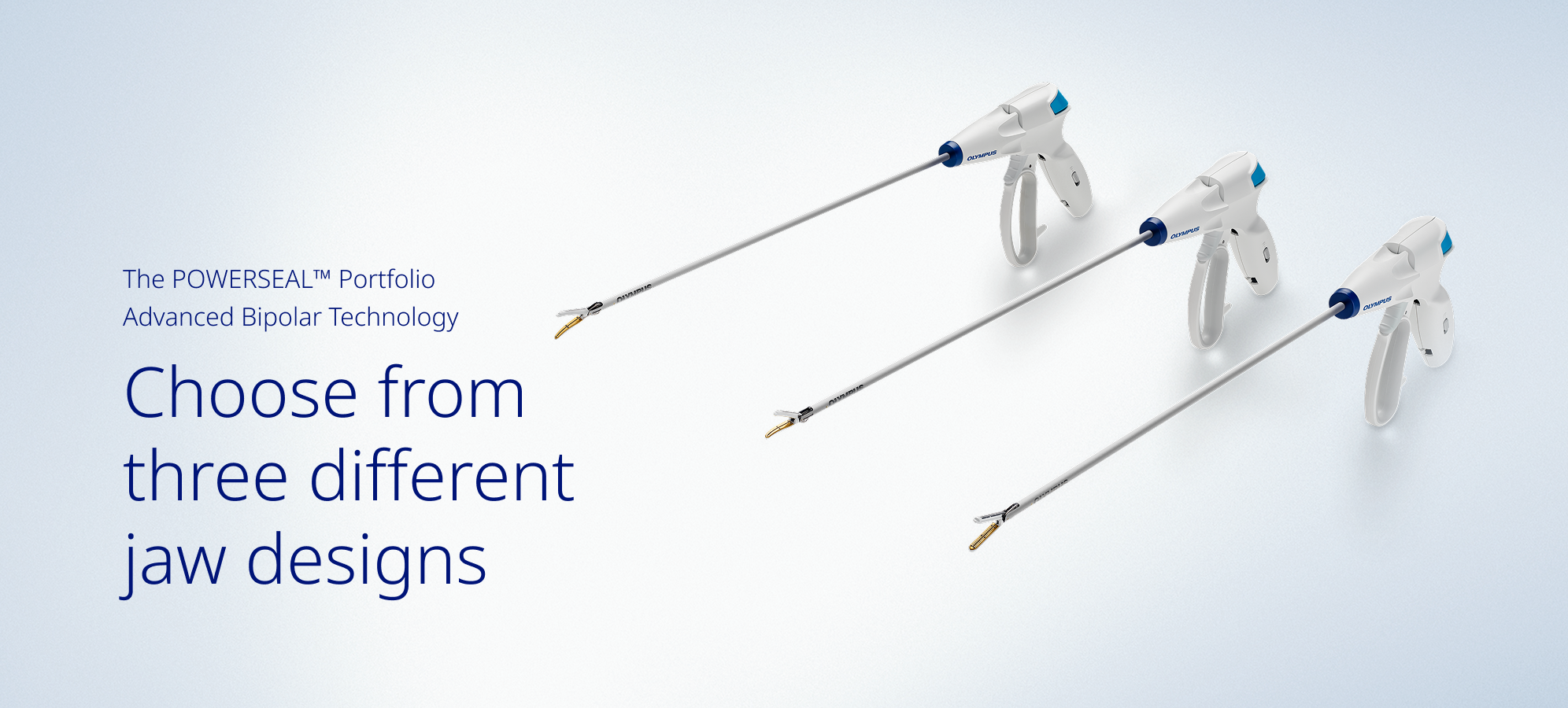
Multifunctional Design
Engineered to accommodate surgeon preference and technique with three jaw styles and one intelligent handle design across the portfolio.
- Three different jaw options to support your preference and technique for dissection, grasping and sealing.
- Latch-on/latch-off setting provides adjustable control to suit user needs.
- Energy activation without requirement to fully close the jaws.7
- Enhanced dissection capabilities8 with greater grasping and dissection force.2
Download the POWERSEAL™ Sealer/Divider brochure to learn more today
Stay informed on the latest POWERSEAL™ Portfolio and Advanced Energy Solutions
The POWERSEAL™ Sealer/Divider is a bipolar electrosurgical instrument indicated for use in a variety of procedures and specialties and can be used on vessels up to and including 7 mm, lymphatics, and tissue bundles. The POWERSEAL Sealer/Divider is intended for single use only and has not been shown to be effective for tubal sterilization or tubal coagulation for sterilization procedures. Consult with a qualified professional prior to utilizing on patients who have electronic implants to avoid possible hazard.
- Data on file with Olympus (DN0054737, DN0054976, DN0046457)
- Data on file with Olympus (DN0042457, DN0046457)
- Data on file with Olympus (DN0056239, DN0056240, DN0056241, DN0056242, DN0044249, DN0044289)
- Data on file with Olympus (DN0046457, DN0056930, DN0044249, DN0044289, DN0045994)
- Data on file with Olympus (DN0046457, DN0056930, DN0044287, DN0044290, DN0049019)
- Data on file with Olympus (DN0046457, DN0056930, DN0044404)
- Data on file with Olympus (DN0024822)
- Data on file with Olympus (DN0055950, DN0044691, DN0049019)